How To Clean With Rubbing Alcohol 70 Or 91
In today's post I'k going to testify you the exactly how to use isopropyl alcohol (IPA) the right manner to disinfect surfaces from 99.99% of microbes including COVID-19.
In fact, this is the same process I use to safely disinfect using isopropyl booze.
And then if you would like to know "How to disinfect with isopropyl alcohol?", you'll love this guide.
Let's jump into it!
How to Utilise Isopropyl Alcohol the Correct way
Isopropyl alcohol (IPA), also known as two-propanol, is a staple cleaning agent. It is the active ingredient in many household cleaners.
It comes diluted with purified h2o or in sanitizer solutions. Typically, to a concentration of 70% to 99% concentration.

Sanitizers (paid link) are commonly made with purified water, glycerine, carborner thickening amanuensis, and fragrance. Some hand sanitizers (paid link) too accept added vitamin Eastward, and aloe vera.

Alcohol-based sanitizers tin can inactivate microbes. But a common mistake is relying simply on booze-based sanitizers to disinfect.
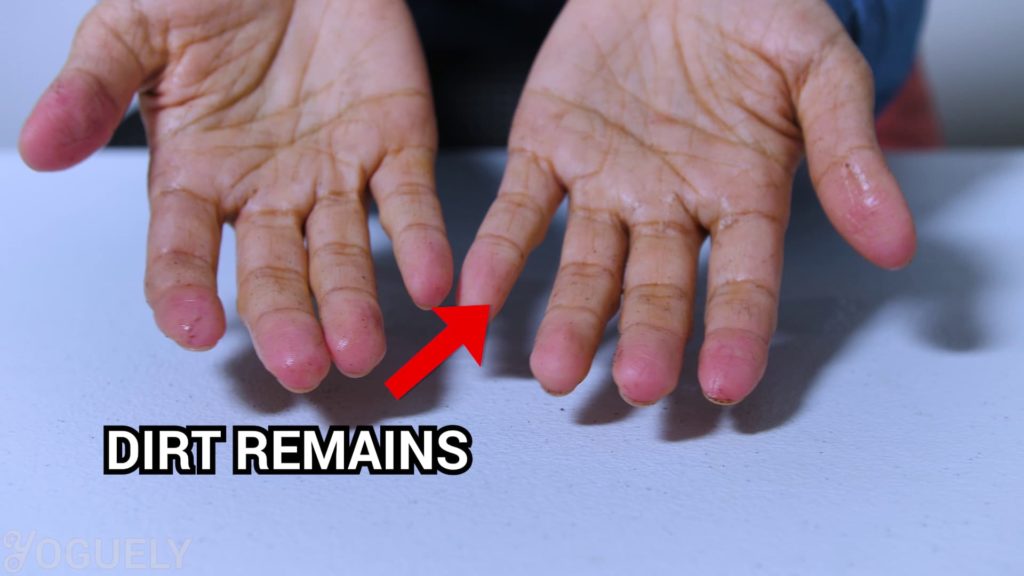
If y'all tin can, wash off the clay and grease first.
Use lather and h2o for your hands or detergent for the surface you are cleaning. That style you reduce the number of microbes. Plus remove certain ones that alcohol cannot.[17]

Then utilise the booze-based sanitizer afterwards to kill whatever remaining germs.

What is Isopropyl Alcohol Skillful For?
Material it helps remove: In terms of materials, isopropyl booze (paid link) is very effective at dissolving oils.
Surfaces you can make clean: Isopropyl alcohol evaporates really quickly, eliminating almost all traces of oil. Given that fact, it is commonly used for metal, plastic, and glass materials.

For case, you can utilize it for cleaning eyeglasses. It is safe to utilize for electronics. Such as: electrical contacts, sound or video tape heads, and optical disc lenses like DVDs.
Information technology is also useful for removing thermal paste from heatsinks on CPU's. Which is something I practise one time a yr on my desktop PC build.

Is 70% Isopropyl Booze a Disinfectant?
70% Isopropyl alcohol is effective in killing 99.99% of germs.
Specifically, isopropyl alcohol diluted with 30% to 10% purified water is a rapid acting bactericidal. Meaning it's capable of killing bacteria. It is also tuberculocidal, fungicidal, and virucidal.
Unfortunately though, they do non destroy bacterial spores.[two] Which explains why solutions of alcohol are non approved by the Food and Drug Assistants (FDA) as a high-level disinfectant.[two]
How does alcohol kill germs?
Alcohol is an antimicrobial solution that works by denaturing the proteins of germs.[2] This in a manner, is similar to how soap works. Alcohol unfolds proteins in living cells, which inactivates cell activeness or kills the jail cell completely. This process is called denaturation.[three]
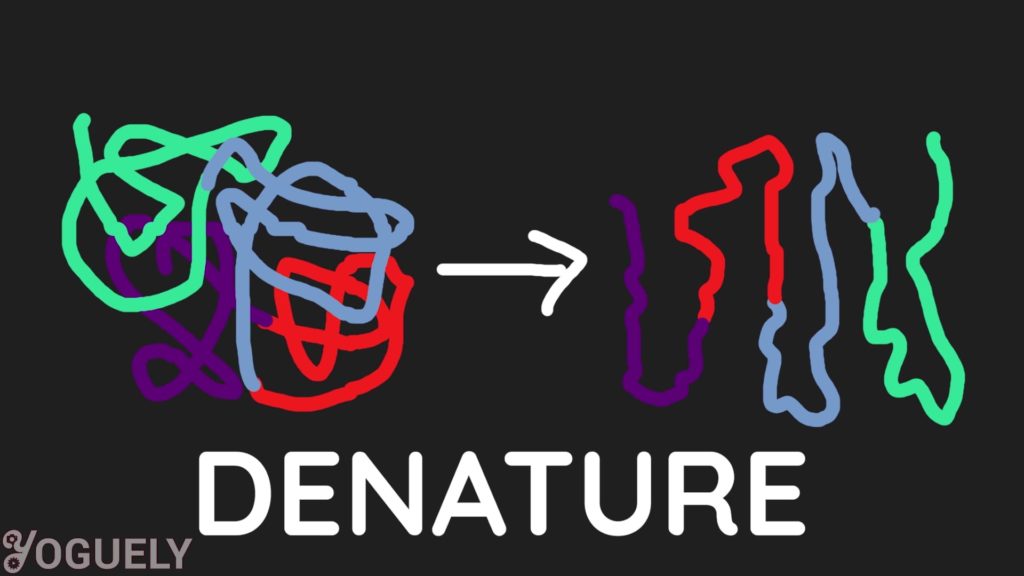
Mixtures of alcohol and purified h2o are more capable of killing bacteria than absolute alcohol. This is because proteins are denatured more rapidly when at that place is water present.[4],[5]
I'll explain this is more detail later on. For now you must be wondering:
Can 70% Isopropyl Alcohol Kill Coronavirus?
Isopropyl alcohol is fully agile against lipid viruses.[seven] Those are viruses who's jail cell membrane are made of a lipid bilayer. In turns out, coronaviruses have a lipid bilayer. Therefore, isopropyl alcohol is very effective in inactivating COVID-nineteen.
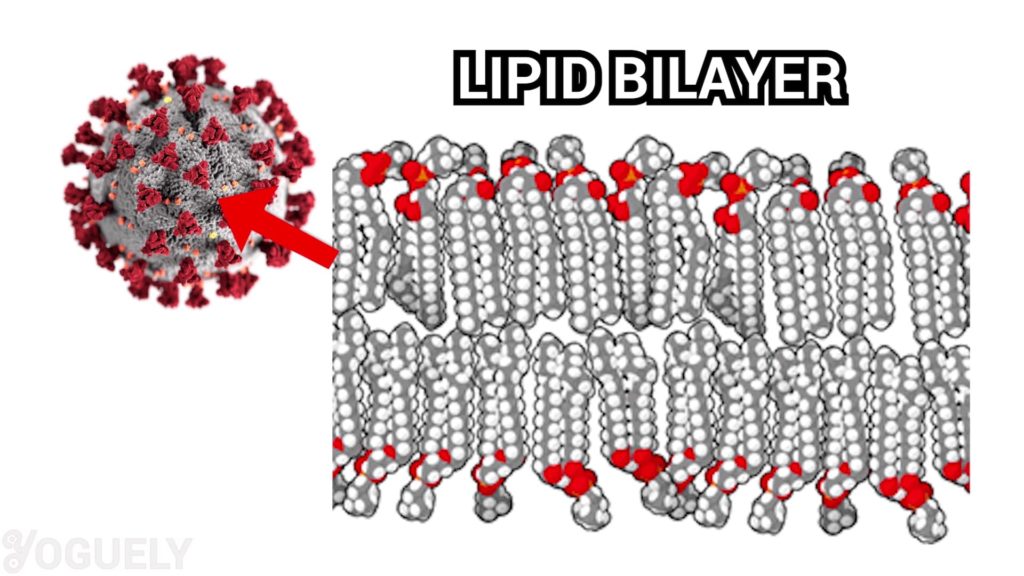
For this reason, the Environmental Protection Agency (EPA) registers the active ingredient isopropyl alcohol as an effective disinfectant for SARS-CoV-2 (COVID-19).[32] Specifically, the CDC too recommends using at least lxx% booze to disinfect at dwelling house.[8],[33]

Other useful disinfectants that can exist used against coronavirus are sodium hypochlorite (bleach), or hydrogen peroxide.
How Long Should lxx% Isopropyl Booze Sit on Surface to Disinfect?
According to the EPA, the contact time is the amount of fourth dimension the surface should be visibly wet.[32]
Contact time: The contact fourth dimension yous need may vary depending on the the microbe y'all wish to destroy. Information technology besides depends on the concentration of isopropyl booze used. For more info, bank check out the lists of EPA-Registered Disinfectants for common pathogens.[34]
I'll exist focusing on COVID-19, since it is one of the biggest threats to our well-being we confront today.
The EPA recommends as much equally five minutes of contact time with solutions of 70% isopropyl booze and 30% water to be effective against COVID-19.[32]
But hold on,
How Long Does It Take for seventy% Isopropyl Alcohol to Evaporate?
Alcohols evaporate VERY rapidly. How fast, will depend on the air substitution rate, and the temperature.
Alcohol's fast dry out times makes information technology difficult to expose a surface to extended contact time.[xvi] Unless, you immerse the surface completely nether booze for the entire v minutes.
You could also experiment by drenching the surface in a thick layer of the liquid. Then measuring how long it takes to dry.
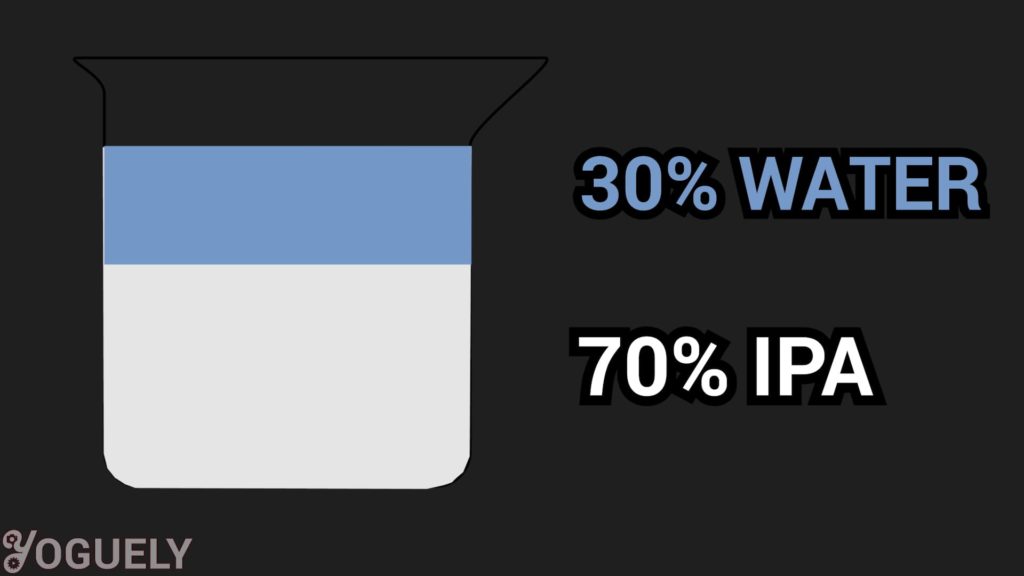
70% vs 91% vs 99% Isopropyl Alcohol (The Deviation)
Y'all've probably seen iii concentrations of diluted isopropyl alcohol being sold. The divergence is the ratio of isopropyl booze to purified water:
- seventy% isopropyl booze and 30% purified h2o
- 91% isopropyl alcohol and 9% purified water
- 99% isopropyl alcohol and one% purified water

Which is amend for disinfecting 70%, 91%, or 99% isopropyl alcohol?
Hither are 3 reasons why 70% isopropyl alcohol is, in most cases, the optimal concentration you will e'er need at dwelling.
- Offset, information technology is longer lasting.
- Second, information technology is more than constructive as a disinfectant.
- Third, it is safer to apply and store.
It Is Longer Lasting.
Alcohol evaporates very rapidly, much faster than h2o. Mixing alcohol with water actually slows down the evaporation rate. So every time you open up and close a container of alcohol, less of information technology volition be used up.
There is some other bigger reward though. By evaporating much slowly, 70% isopropyl booze sits on surfaces for longer. Which, increases the contact time, and hence its effectiveness.
Information technology Is More than Effective as a Disinfectant.
Another key point, we are looking for the all-time bactericidal. Like I mentioned before, alcohol and water is more than effective every bit a bactericidal than alcohol alone.
Now here is the interesting fact. Enquiry shows that equally you dilute alcohol with purified h2o below 50% alcohol, the solution becomes less constructive. The optimal concentration is really between 60% and ninety% alcohol.[2],[iv],[6]
Particularly, 70% isopropyl alcohol lands within the optimal concentration range.
It Is Safer to Utilise and Store.
Isopropyl alcohol is highly combustible. The higher the concentration on alcohol, the more than dangerous information technology is as a flammable solution. So y'all don't desire to exist keeping high concentrations of information technology lying around.
For your rubber at home, apply the least concentration of isopropyl alcohol that is still constructive against the microbe y'all want to eradicate.
70% isopropyl alcohol is the well-nigh diluted solution that nevertheless lies within the optimal concentration range recommended by the CDC.
For these three reasons: stability, effectiveness, and safety. And because of my experience in engineering applications. I become with 70% isopropyl alcohol whenever disinfecting at habitation.
Isopropyl Booze Safety Measures
Here are some safety measure out to have whenever working with isopropyl alcohol.

MSDS: Safety Data Sheet for 70% Isopropyl Booze
Hazards: As far every bit hazards become, isopropyl booze is highly flammable. It is also an irritant so go on information technology away from your eyes and skin.

Some other key point, alcohol evaporates apace. And if you jiff it, it can make you feel drowsy or giddy.
Personal Protective Equipment (PPE): To prevent those hazards when handling isopropyl alcohol, you should wear personal protective equipment (PPE).
First off, wearable protective clothing and gloves (paid link) to cover your skin. As important, wear chemical safe goggles (paid link) that meets the OSHA'southward protection regulations.[eighteen],[nineteen]
In similar mode, wear respiratory protection (paid link) by the NIOSH or the MSHA.[10]

Have an emergency eye wash and safe shower nearby.

Also, utilize an exhaust ventilation to keep airborne concentrations low. Remove all possible sources of ignition. Besides, all your electrical equipment should be explosion-proof.
Key betoken frequently overlooked, take precautionary measures against static discharges.

If you are using booze on your hands, make certain your hands are completely dry. So that all the alcohol has evaporated before you impact anything. Whatsoever small static accuse could spark a fire, and cause serious burns.
Shelf Life of Isopropyl Alcohol
Shelf Life: A 70% isopropyl booze product will take a EXP expiration appointment on printed on information technology which is nearly 3 years from the date you buy information technology. Afterwards 3 years, the production might still be usable every bit long as the cap was sealed well after every use.
Here is why:
If yous don't seal the cap well, alcohol will brainstorm to evaporate much faster than h2o.
Specifically, the boiling bespeak of isopropyl alcohol is 82.6 °C (180.seven °F; 355.8 1000). Which, is much lower than the boiling point of water 100 °C (212 °F; 373.15 G).
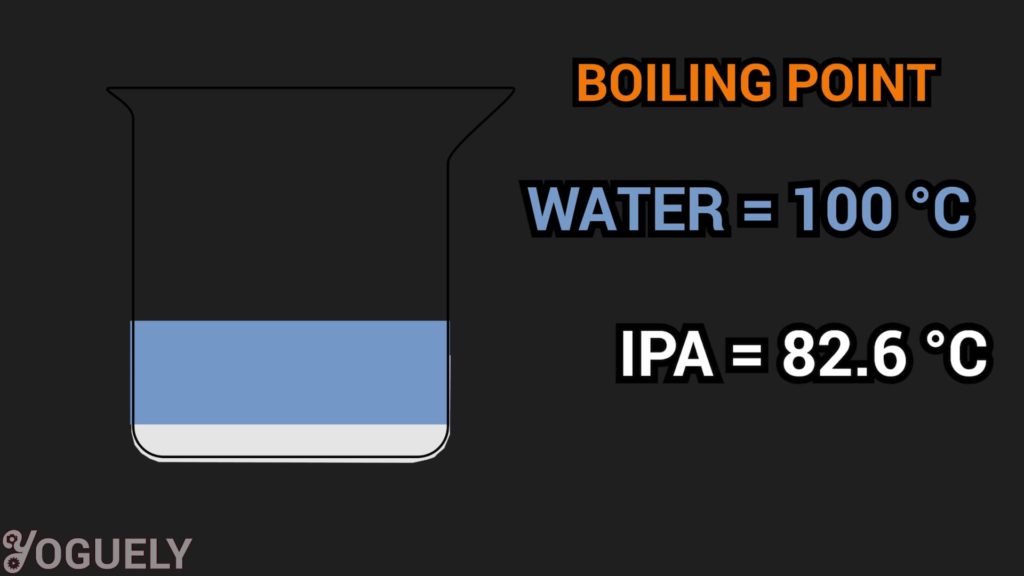
Over fourth dimension, the concentration of booze will exist much less, and the solution that remains will only contain h2o.
Besides information technology's effectiveness, there is some other thing y'all need to consider. That is, the danger of using isopropyl booze that has been exposed to air and then stored for a long time.
Isopropyl alcohol is a peroxide forming chemic. And then information technology will react with air or oxygen to course unstable peroxides. These peroxides could accrue at extremely unsafe concentrations. If that happens, information technology can crusade an explosion.[11],[12],[13]

In fact, there already has been explosions in lab experiments due to peroxide formation from isopropyl alcohol.[14],[fifteen]
So information technology may be a adept idea to only employ up the entire bottle before its expiration engagement to avert having a bad twenty-four hour period.
How to Shop Isopropyl Alcohol for Long Term
Alcohols should be stored in a fire resistant or preferably a fireproof storage.[nine] That storage should exist located in a absurd, well-ventilated area, and away from oestrus sources that may cause a burn.
Alcohol is probably the most common flammable chemical I've worked with at NASA. I recall e'er storing it in a fire rated chemical storage cabinets. Kind of like this combustible's mini safety storage cabinet (paid link), but style bigger.

Store alcohol separately from other chemicals that could react adversely. Peculiarly since information technology is combustible.
So you'll want to keep information technology abroad from strong oxidants like hydrogen peroxide.
Remember to never mix alcohol with bleach. Considering it forms chloroform, a toxic coldhearted and sedative.

Conclusion
There you take information technology folks. That's my guide on how to utilise isopropyl alcohol the right way.
Exit your thoughts in the annotate department below. Or join the discussion in the Yoguely Community Forum.
I'thou Aida Yoguely. Thanks for learning with me today. And I'll see you shortly.
To stay tuned, join our newsletter and get the latest content direct to your inbox.
Video
How To Use Isopropyl Alcohol Disinfectant (70% vs 91% vs 99%)
Be sure to subscribe and hitting the notification bell to stay tuned for the latest videos.
References
1. ^ (May 28, 2022). "Isopropyl alcohol". Wikipedia. Retrieved June 1, 2022.
2. ^ (Sept xviii, 2022). "Guideline for Disinfection and Sterilization in Healthcare Facilities (2008)". CDC. Retrieved May xxx, 2022.
three. ^ (May 8, 2022). "Denaturation (biochemistry)". Wikipedia. Retrieved May 23, 2022.
4.^ Ali Y, Dolan MJ, Fendler EJ, Larson EL. Alcohols. In: Cake SS, ed. Disinfection, sterilization, and preservation. Philadelphia: Lippincott Williams & Wilkins, 2001:229-54.
v. ^ Morton HE. Alcohols. In: Cake SS, ed. Disinfection, sterilization, and preservation. Philadelphia: Lea & Febiger, 1983:225-239.
half-dozen. ^ Morton HE. The relationship of concentration and germicidal efficiency of ethyl alcohol. Ann N.Y. Acad. Sci. 1950;53:191-96.
7. ^ Klein M, DeForest A. The inactivation of viruses by germicides. Chem. Specialists Manuf. Assoc. Proc. 1963;49:116-viii.
8. ^ (April ii, 2022). "Cleaning And Disinfecting Your Domicile". CDC. Retrieved June one, 2022.
9. ^ "Laboratory Chemic Condom Summary (LCSS) Isopropyl booze". National Library of Medicine. Retrieved June ane, 2022.
10. ^ (January 1996). "NIOSH Guide to the Option and Use of Particulate Respirators". CDC. Retrieved May 27, 2022.
eleven. ^ "Compound Summary: Isopropyl alcohol". National Library of Medicine. Retrieved June 1, 2022.
12. ^ "Peroxide Formation in Chemicals". UC Davis. Retrieved June 1, 2022.
13. ^ Clark, D. Eastward. (2001). Peroxides and peroxide-forming compounds. Chemic Wellness and Rubber, 8(5), 12-22.
fourteen. ^ Mirafzal, G. A., & Baumgarten, H. Due east. (1988). Control of peroxidizable compounds: An addendum. Journal of Chemic Education, 65(9), A226.
15. ^ (Aug ane, 2022). "Chemic safety: peroxide formation in 2-propanol". Chemical & Engineering News. Retrieved June 1, 2022.
16. ^ Venkataram, M. (2012). Textbook on Cutaneous and Aesthetic Surgery. JP Medical Ltd.
17. ^ (March three, 2022). "Show Me the Science – When & How to Use Hand Sanitizer in Community Settings". CDC. Retrieved June ii, 2022.
eighteen. ^ "Centre and Face protection". United States Department of Labor Occupational Safety and Health Administration. Retrieved June 6, 2022.
nineteen. ^ "Selecting PPE for the Workpace". United States Department of Labor Occupational Rubber and Health Administration. Retrieved June vi, 2022.
32. ^ "List N: Disinfectants for Use Confronting SARS-CoV-ii". EPA. Retrieved May 27, 2022.
33. ^ (July ten, 2022). "Cleaning And Disinfection for Households". CDC. Retrieved September 25, 2022.
- Author
- Contempo Posts
Source: https://www.yoguely.com/isopropyl-alcohol/
Posted by: kingseved1947.blogspot.com


0 Response to "How To Clean With Rubbing Alcohol 70 Or 91"
Post a Comment